  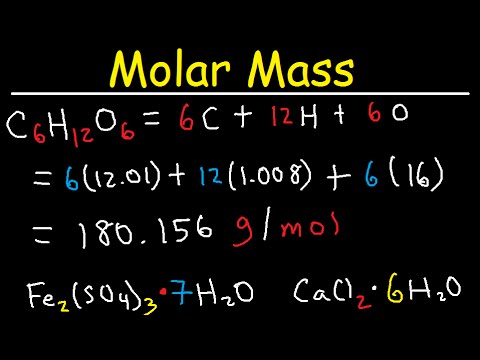
Compute the value of percent recovery using the formula below.When calculating molecular weight of a chemical compound, it tells us how many grams are in one mole of that substance. Finding molar mass starts with units of grams per mole (g/mol). Weigh the dried substance and record the value. Using the chemical formula of the compound and the periodic table of elements, we can add up the atomic weights and calculate molecular weight of the substance.On purifying the desired material, leave it aside to dry.Weigh the original amount of the substance.While percent recovery is calculated as the ratio between the pure compound and initial compound, percent yield is calculated as a ratio between actual yield and theoretical yield. Most people tend to mix up percent recover and percent yield. Percent recovery is very important for industries trying to make the most product with the least waste.We use percent recovery to determine the efficiency of a purification process.Percent recovery is used in cases where no chemical reaction is taking place, as in purification of a sample.This is a process where a chemical is dissolved in a hot solvent and then precipitated out again by cooling the solution, leaving impurities behind.Īdditionally, based on Vogel’s Textbook of Practical Organic Chemistry, yields near to 100% are known as quantitative, yields above 90% are known as excellent, yields above 80% are extremely good, yields above 70% are great, yields above 50% are fair, and yields below 40% are known as poor. The percent recovery concept is often used in organic chemistry when dealing with the recrystallization processes. 
It is normally expressed as a percentage value. With that in mind, we can simply define percent recovery as the the amount of pure compound with respect to the impure compound obtained after a purification process. During these procedures, some of the required product may be lost and this results in a reduced recovery. So he does a purification procedure to recover the lost product. Formular Weight Calculator Putting in a molecular formula of any type such as K2Cr2O7, CH3CH2COOH, KFe Fe (CN)63, or Na2B4O7.10H20 will result in the molar mass and mass analyses being calculated when the button is pressed. Sometimes a Chemist in the lab may perform a procedure where some chemical product can be lost. 06 All right, So for that I am frequent formula.So as to calculate percent recovery, it is essential for us to know its meaning and where this concept is used. So the market the formula will be ah si six C six h 12. So we're coming Teoh 100 into also Jinyu goes to 6 to 12 to six. I hope that students will find this useful for double checking their homework problems and. I am a former Organic Chemistry student who created an Organic Chemistry calculator to help solve 1-step synthesis reactions: I am trying to build a tool that helps students who are studying Ochem remotely. So ah, we're going to file them while conforming formula by extend bishop more away show which in carbon, hydrogen and oxygen. Organic Chemistry Reaction Calculator Tool. Home » Chemistry » Compound Name and Formula Search. Okay, so I'm going to convert all filth alien to to do them bone Most so for ah, cabinet were six more on Dan for hydrogen would be rough, arrive on 12 and therefore sausage whoever around six as well. Chemical Compounds Lookup by Name or Formula. And then that's been known this for our situation. It took the author of the article several hours to balance this equation manually, and another hour to check the answer. Um, Graham And then for oxygen, we will have Ah, this is from 7% time 1 80.1 so ever around 11.1 cram. Here it is: Cr (N 2 H 4 CO) 6 4 Cr (CN) 6 3 +KMnO 4 +H 2 SO 4 K 2 Cr 2 O 7 +MnSO 4 +CO 2 +KNO 3 +K 2 SO 4 +H 2 O. How for your sentence will be over for around 72. So for the 1st 1 we would have won 84 in one time. So, first off, I would convert, um, the percentage, um, to master using them with a mass. So we're giving the other man's off compound and also the mass membership we're gonna found them. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
